Mum welcomes new drug laws

Doctors in the UK will now be able to prescribe cannabis-derived medicine after the Government last week announced a relaxation of laws governing access to the substance.
Thousands of people with drug-resistant conditions will potentially be able to use cannabis-derived medicinal products for treatment after the Home Secretary, Sajid Javid, announced they should be placed in schedule 2 of the 2001 Misuse of Drugs Regulations, allowing clinicians to prescribe them by the autumn.
Advertisement
Hide AdAdvertisement
Hide AdCannabis has been classed as a schedule 1 drug, meaning it is thought to have no therapeutic value and cannot be lawfully possessed or prescribed. It may be used for the purposes of research, but a Home Office licence is required.
The Department of Health and Social Care and the Medicines and Healthcare Products Regulatory Agency will now develop a clear definition of what constitutes a cannabis-derived medicinal product so they can be rescheduled and prescribed. Only products meeting this definition will be rescheduled.
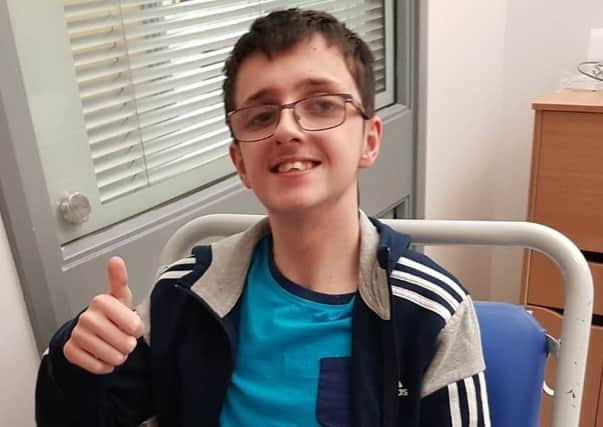
Now, Amble mum Sarah Nicholson, whose 16-year-old son Brandon was diagnosed with an inoperable brain-stem tumour when he was two and had surgery this year to remove a growth from his spine, has welcomed the move.
She said: “I’ve been a supporter of the legalisation of cannabis for many years.
Advertisement
Hide AdAdvertisement
Hide Ad“I feel Brandon – and so many other people – would benefit from cannabis oil, easing his pain and side effects.
“It is amazing that so many people are realising the medicinal benefits cannabis does have and I’m hoping things are finally moving in the right direction in the UK. I would love to let Brandon be able to have access to the oil without that fear of breaking the law.”
Wansbeck MP Ian Lavery also welcomed the news, having submitted a parliamentary petition on behalf of constituent Paul Keeney, who suffered cancer and called for the Government to legalise medical cannabis for patients.